  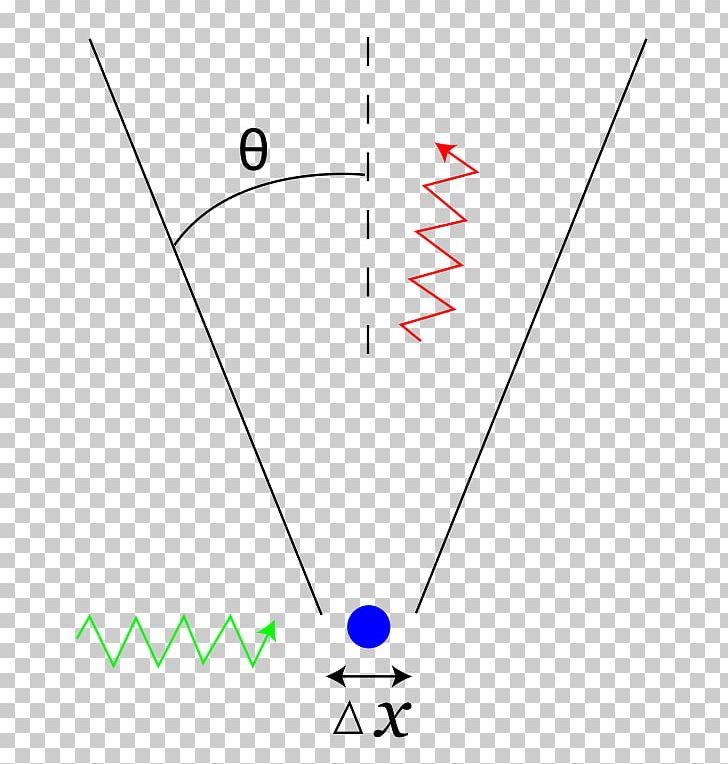
But the act of measurement involves interacting with the electron, which will cause it to change its momentum, increasing your uncertainty about that value. 
Instead, they are more like clouds of probabilities, with a varying chance of appearing in a certain place, which gives them a wave-like nature.Īs you repeatedly measure an electron, you can build up certainty about its position. Particles governed by quantum physics, such as the electron, don’t really exist in a specific place. It’s a concept that makes no sense on a human scale, but that’s quantum mechanics for you. Informally, this means that both the position and momentum of a particle in quantum mechanics can never be exactly known. The two error sizes, when multiplied together, will always exceed a certain value. It states that any effort to reduce the error in your measurement of a particle’s momentum (that is, its mass multiplied by its velocity) will increase the error in your measurement of its position. This bizarre trade-off arises from Heisenberg’s uncertainty principle, which was first laid out by German physicist Werner Heisenberg in 1927. For quantum particles, this is a reality: the better you know a particle’s speed, the less certain you are of its position. l is intuitively related to the total angular momentum (how quickly the electron rotates) and m is related to the direction of the motion.Imagine driving a car fitted with a GPS navigation system that glitches every time you look at the speedometer. Recall we introduced the p, x uncertainty principle by. At this point, the analogy with p x emerges. We can then achieve an intuitive understanding of the quantum number l and m which appears in the free rotor and the hydrogen atom eigenfunction. This is a far slower time dependence than that of the e iE0t / term, so it is an excellent approximation to put the time dependences together in one exponential factor: (t) (0)e i ( E0 i / 2) t /. The Heisenberg uncertainty principle x × p h tells us that whenever we try to measure certain pairs of numbers we cannot get both values with high accuracy. Once the uncertainty in momentum p p is found, the uncertainty in velocity can be found from p mv p m v. Thus the smallest uncertainty in momentum p p can be calculated using xp h/4 x p h / 4. The principle that it is not possible to know with unlimited accuracy both the position and momentum of a particle. Can you also have a precise value for a different quantity B? The answer is generally no, because the eigenfunctions of A ^ (Eq. The uncertainty in position is the accuracy of the measurement, or x 0.0100 nm x 0.0100 n m. In quantum mechanics, the Heisenberg uncertainty principle states that there will always be some amount of uncertainty involved in making measurements and. If we measure A, we will always get a, i.e. However, it typically assumes that we probe these properties via measurements at a single point in time. The uncertainty principle, originally formulated by Heisenberg 1, clearly illustrates the difference between classical and quantum mechanics.The principle bounds the uncertainties about the. Suppose that we have prepared a system in a state φ and this state is an eigenfunction of the operator  corresponding to a classical quantity A ( Âφ = aφ). Heisenbergs uncertainty principle implies fundamental constraints on what properties of a quantum system we can simultaneously learn. 
Type classification: this is a lesson resource. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
